The Fujifilm Group's healthcare business is engaged in a wide range of areas, encompassing prevention, diagnosis, and treatment as a total healthcare company. FUJIFILM Toyama Chemical, one of the core companies in the healthcare segment, is committed to solving social issues through its pharmaceutical business.
Leveraging over 40 years of expertise in anti-infective drugs, we have recently focused on contract manufacturing as the only company capable of manufacturing sterile penicillin antibiotics products in Japan.
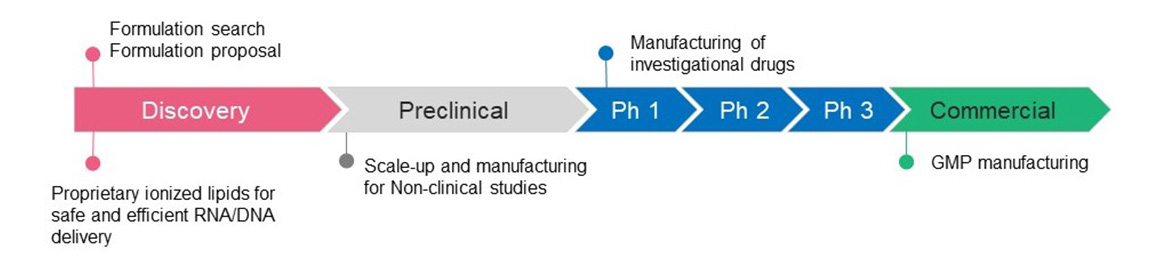
In addition, we are advancing our CDMO*1 business for formulations using lipid nanoparticle (LNP), liposome, and other drug delivery system (DDS) technologies, which are also used in the mRNA vaccines for COVID-19. A new formulation manufacturing facility capable of producing commercial products became operational in 2020. Going forward, we will expand the CDMO business, including new modalities such as LNP, to meet customer needs.
Furthermore, FUJIFILM Toyama Chemical has been offering tailored drug discovery services since 2023. Our drug discovery services are structured to expedite our clients' drug discovery and development processes by utilizing our advanced research platform and extensive expertise, in addition to collaborating with our CDMO team.
- *1 CDMO stands for Contract Development and Manufacturing Organization.
We are promoting the development of drugs for drug-resistant bacteria, so-called “silent pandemics,” and for diseases caused by highly lethal and virulent viruses.
As the only manufacturer in Japan capable of synthesizing penicillin antibiotics and producing sterile products, we also engage in contract manufacturing.
We deploy an integrated CDMO business from the design to process development and GMP production of formulations using LNP and liposomes, which are DDS technologies, by leveraging Fujifilm’s advanced nano dispersion, analysis, and synthesis technologies cultivated and evolved through the development of a wide range of products including photographic films, as well as our production expertise and manufacturing facilities from 2020.
We will begin offering a mRNA CDMO service from fiscal year 2023.
In order to respond to the wide-ranging needs of our customers, CDMO services for antibodies*2 and ADC*3 (Antibody Drug Conjugate) will be launched in near future.
FUJIFILM Toyama Chemical Co., Ltd. supports customers one-stop from research stage to commercial manufacturing in formulation design, formulation optimization and manufacturing.
- *2 Our group company FUJIFILM Biotechnologies is responsible for the development and contract manufacturing of antibody pharmaceuticals.
- *3 ADC process/analytical method development and small-scale non-GMP manufacturing services are scheduled to begin in the first half of FY2025, and GMP manufacturing will commence in the second half of FY2027.
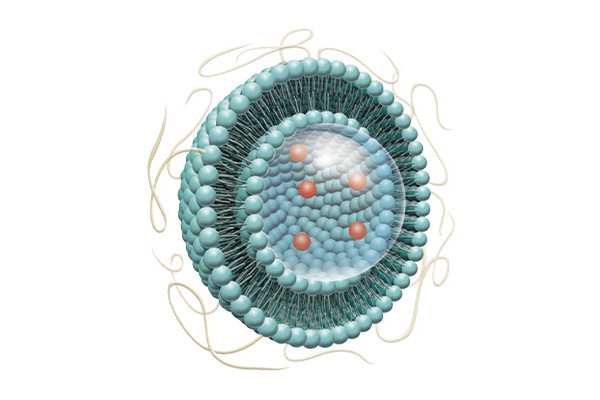
Formulation of low-molecular-weight compounds encapsulated in lipid bilayer
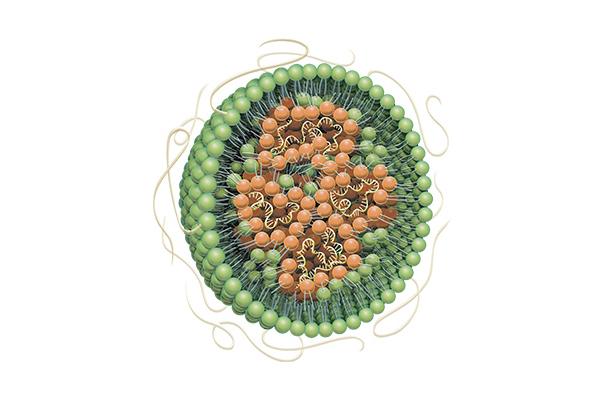
Formulation in which nucleic acids such as mRNA are encapsulated in lipid nanoparticles

There is active develop as treatments for cancer, infectious diseases, genetic diseases, etc.
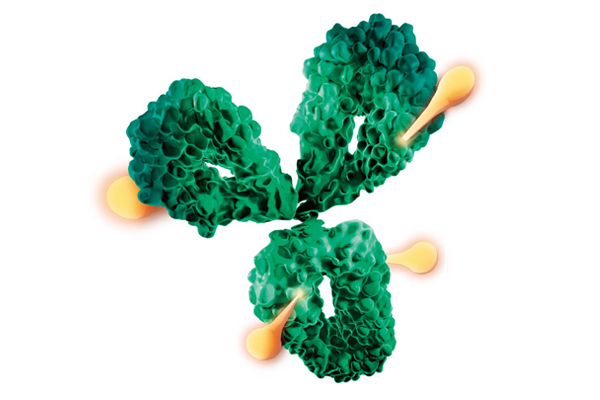
This biopharmaceutical combines a drug with an antibody to selectively target disease sites.
FUJIFILM Toyama Chemical has been offering tailored drug discovery services since 2023. With our strong, over a decade-long history of identifying various anti-infectives ranging from target identification to IND submission, we have developed a state-of-the-art platform aimed at discovering innovative treatments for severe, life-threatening infectious diseases. Our team of experts brings a wealth of knowledge across diverse drug discovery modalities, including small molecules, natural products, peptides, antibody-drug conjugates, and vaccines. Furthermore, our drug discovery services are structured to expedite our clients' drug discovery and development processes by utilizing our advanced research platform and extensive expertise, in addition to collaborating with our CDMO team.
