TOKYO, May 8, 2026 - FUJIFILM Corporation (President and CEO, Representative Director: Teiichi Goto) announces that its group company, FUJIFILM Toyama Chemical Co., Ltd. (President: Mitsuhiro Sato), has received manufacturing and marketing approval in Japan for SAVYSCUS® Injection, a regenerative medical product. It is indicated for meniscal injuries for which meniscectomy is considered appropriate and is the first regenerative medical product approved in Japan*1 for meniscal injury under the Pharmaceutical and Medical Device Act.
SAVYSCUS® Injection is composed of autologous synovial mesenchymal stem cells that are isolated and expanded from synovial tissue harvested from the patient’s own knee joint. Traditionally, approximately half of meniscal injuries have been considered indications for meniscectomy, in which the damaged portion of the meniscus is removed. While this procedure may alleviate symptoms, it has been reported that removal of the meniscus may increase the long-term risk of progression of knee osteoarthritis*2. As a result, there has been growing interest in treatment strategies that emphasize preservation of the meniscus in orthopedic practice. With this approval, SAVYSCUS® Injection provides a new treatment option as a regenerative medical product for patients with meniscal injuries for whom meniscectomy would otherwise be considered, in line with meniscus-preserving treatment concepts.
The approval was based on the results of a feasibility clinical trial, which is led by Professor Ichiro Sekiya, Director of the Center for Stem Cell and Regenerative Medicine, Institute of Science Tokyo, and a pivotal Phase III clinical trial. FUJIFILM Toyama Chemical submitted the application for manufacturing and marketing approval on May 13, 2025, which has now been approved. The Phase III study was a single-arm, multicenter clinical trial enrolling 19 patients with meniscal injuries indicated for meniscectomy. The primary endpoint was the change in the Lysholm knee score*3 from screening to 52 weeks after administration.
Additionally, to objectively assess the repair of meniscal lesions before and after administration, arthroscopic evaluation by an independent expert was conducted at week 52 post-administration as secondary endpoint. Furthermore, follow-up MRI evaluations were performed up to 104 weeks after administration.
In the orthopedic field related to knee joint disorders, Fujifilm also provides a knee joint analysis software application as part of its 3D image analysis system, SYNAPSEVINCENT.®*4 This software has been utilized in epidemiological studies on knee osteoarthritis, such as the Kanagawa Knee Study and its longitudinal follow-up study*5. This software is expected to contribute to the diagnosis of knee osteoarthritis and meniscal injuries, which is considered early-stage pathology of knee osteoarthritis.
The name SAVYSCUS® Injection is derived from the internationally recognized concept “Save the Meniscus,” reflecting the philosophy of meniscus preservation. Through SAVYSCUS® Injection, the Fujifilm Group aims to provide new treatment options for meniscal injury and to contribute to improving and maintaining human health by offering a broad range of orthopedic solutions spanning prevention, diagnosis, and treatment.
- *1 SAVYSCUS® Injection is the first regenerative medical product approved in Japan for meniscal injury under the Pharmaceutical and Medical Device Act.
- *2 A condition characterized by cartilage degeneration, affecting an estimated over 20 million people in Japan.
- *3 A validated scoring system widely used to assess knee symptoms and function, particularly suited for meniscal disorders.
- *4 SYNAPSE VINCENT® refers to the following medical device: Certification No. 22000BZX00238000
- *5 Led by Professor Ichiro Sekiya, Director of Center for Stem Cell and Regenerative Medicine, Institute of Science Tokyo, with the cooperation of Kanagawa Prefecture.
The meniscus is a C-shaped fibrocartilaginous tissue in the knee joint that plays a critical role in shock absorption, load distribution, and joint stability. Meniscal injuries may occur as a result of aging-related degeneration or sports-related trauma, and are commonly associated with symptoms such as pain, swelling, catching or locking sensation, and instability during knee movement.
Treatment options for meniscal injury include conservative management, meniscal repair, and meniscectomy (partial removal of the meniscus). Because meniscal repair is applicable only to limited injury patterns and locations, meniscectomy is frequently selected for injuries that are unlikely to heal with repair, accounting for approximately half of all meniscal surgeries performed in Japan.
Although meniscectomy can provide symptomatic relief, loss of the meniscal tissue increases mechanical stress on the articular cartilage and has been reported to elevate the long-term risk of progression to knee osteoarthritis. As osteoarthritis progresses, some patients may ultimately require more invasive procedures, such as total knee arthroplasty.
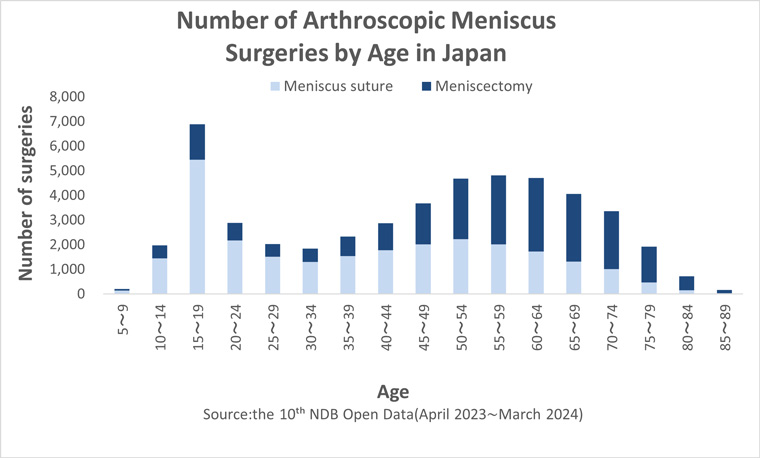
In Japan, approximately 50,000 meniscal surgeries are performed annually, with around half involving meniscectomy, while this number increases to 60% in patients over 40 years old.
SAVYSCUS® Injection incorporates synovial mesenchymal stem cell-based technology developed by Professor Ichiro Sekiya, Director of the Center for Stem Cell and Regenerative Medicine, Institute of Science Tokyo. The therapy involves intra-articular administration of a suspension of cultured autologous synovial mesenchymal stem cells using a minimally invasive arthroscopic procedure. This approach is intended to align with treatment strategies aimed at meniscus preservation in patients with meniscal injuries for whom meniscectomy would otherwise be considered.
Fujifilm and the Institute of Science Tokyo have jointly conducted research on the treatment of meniscal injury using this technology. These research activities were carried out under projects supported by the Japan Agency for Medical Research and Development (AMED) *6, including the feasibility clinical trial*7 aimed at clinical implementation.
- *6 “Practical Application of Regenerative Medicine for Knee Osteoarthritis (Cartilage and Meniscus) Using Synovial Stem Cells” (Supported period: Fiscal Years 2015–2017)
- *7 “Physician-Initiated Clinical Trial of Autologous Synovial Stem Cells for the Treatment of Meniscal Injury” (Supported period: Fiscal Years 2018–2019)
FUJIFILM Toyama Chemical designed and conducted the pivotal Phase III clinical trial based on the results of the preceding feasibility clinical trial. This study was initiated in January 2023 to evaluate the efficacy and safety of SAVYSCUS® Injection in patients with meniscal injuries indicated for meniscectomy. This was a single-arm, multicenter clinical trial enrolling 19 patients, including those with suspected flap tears, who had not achieved improvement following at least three months of conservative treatment.
The primary endpoint was the change in Lysholm knee score from screening to 52 weeks after administration. As a result, the Lysholm knee score (mean ± standard deviation) at screening was 38.1 ± 8.9, and at 52 weeks after the end of treatment, it was 91.6 ± 8.2; the change in the Lisholm knee score (least squares mean [95% confidence interval]) was 53.5 [48.7, 58.2], and a statistically significant difference was observed for this clinically meaningful change. Subsequently, the change in the Lisholm knee score at week 104 was 57.8 [54.4, 61.2], indicating that the improvement in meniscal function and symptoms observed at week 52 was maintained.
Secondary endpoints included arthroscopic image assessments by independent experts at 52 weeks, as well as longitudinal MRI evaluations by independent experts up to 104 weeks after administration. Based on arthroscopic image assessment, complete healing in the central region of the meniscus — an avascular zone where spontaneous healing is not expected — was observed in 35.7% of patients with flap tears (5 of 14 patients*8). In addition, based on MRI evaluation, improvement was observed in 73.7% of all patients (14 of 19 patients).
During the follow-up period, no reoperation related to meniscal re-tear was reported, and no adverse events with a causal relationship to the product were identified.
- *8 In one case, arthroscopy at 52 weeks post-administration was not performed due to the occurrence of an adverse event.
FUJIFILM Holdings Corporation
Corporate Communications Division
Public Relations Group
FUJIFILM Toyama Chemical Co., Ltd.
Product Information Center
FUJIFILM Corporation
Life Sciences Business Division
- * Please note that the contents including the product availability, specification, prices and contacts in this website are current as of the date of the press announcement and may be subject to change without prior notice.




